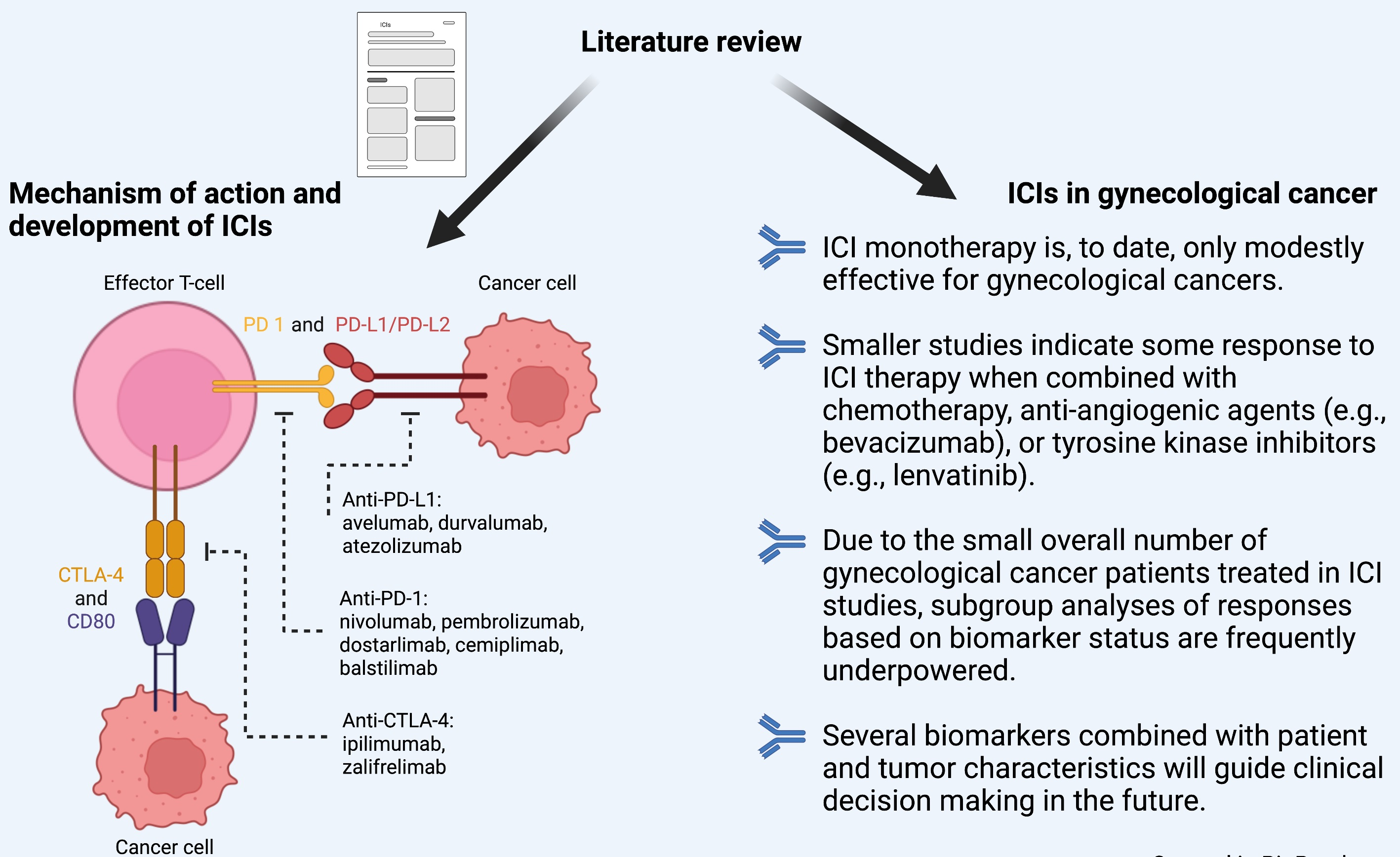
Cancers | Free Full-Text | Overview of Immune Checkpoint Inhibitors in Gynecological Cancer Treatment

Stephen V Liu, MD on Twitter: "#ASCO22 Discussion of PEARLS (KEYNOTE-091, @ETOP_eu 8-15), a phase III study of adjuvant pembrolizumab in resected NSCLC. The study did meet its primary endpoint (DFS HR

KEYTRUDA® (pembrolizumab) Showed Statistically Significant Improvement In Disease-Free Survival Versus Placebo As Adjuvant Treatment For Patients With Stage IB-IIIA Non-Small Cell Lung Cancer Regardless Of PD-L1 Expression 2023 - EORTC

Stephen V Liu, MD on Twitter: "#ASCO22 Discussion of PEARLS (KEYNOTE-091, @ETOP_eu 8-15), a phase III study of adjuvant pembrolizumab in resected NSCLC. The study did meet its primary endpoint (DFS HR

Adjuvant pembrolizumab extends DFS for resectable NSCLC regardless of PD-L1 expression | oncology.medicinematters.com

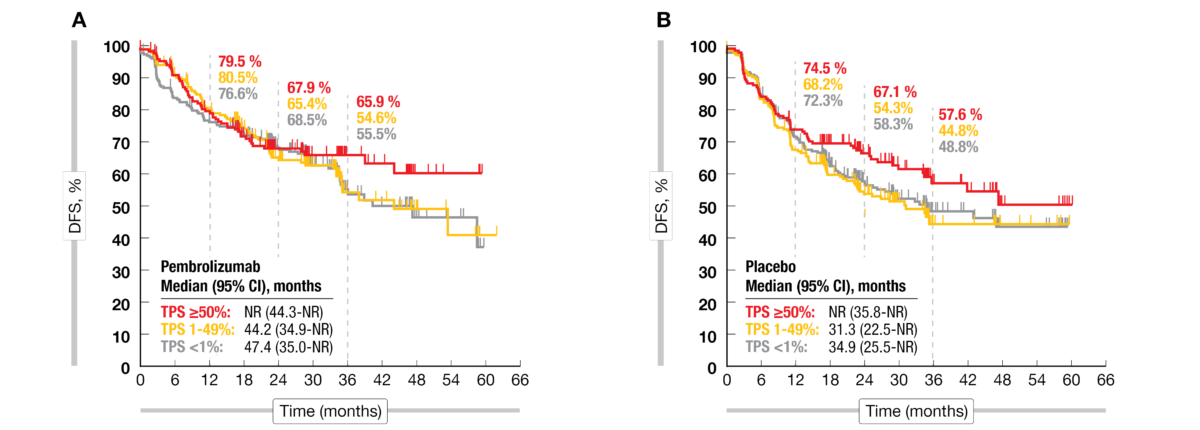
Pembrolizumab versus placebo as adjuvant therapy for completely resected stage IB–IIIA non-small-cell lung cancer (PEARLS/KEYNOTE-091): an interim analysis of a randomised, triple-blind, phase 3 trial | AIOM
Pembrolizumab versus placebo as adjuvant therapy for completely resected stage IB–IIIA non-small-cell lung cancer (PEARLS/KEYNOTE-091): an interim analysis of a randomised, triple-blind, phase 3 trial | AIOM

Schema of the ACCIO trial including three arms. Pembrolizumab dosing is... | Download Scientific Diagram

Aakash Desai, MD, MPH on Twitter: "@LuisPaz_Ares discussed data from #PEARLS study #Pembrolizumab for early stage #NSCLC #LCSM with dual primary endpoint of DFS in all comers/PDL1>50%, showing significant DFS improvement HR:0.76

Benjamin Besse on Twitter: "Adjuvant pembrolizumab improves Disease Free Survival in pts after complete resection of stage IB-IIIA Non-Small Cell Lung Cancer HR=0.76 (IC95 0.63-0.91), including subgroups PD-L1 neg (HR=0.78) or stage

Pembrolizumab versus placebo as adjuvant therapy for completely resected stage IB–IIIA non-small-cell lung cancer (PEARLS/KEYNOTE-091): an interim analysis of a randomised, triple-blind, phase 3 trial - The Lancet Oncology

Pembrolizumab improves DFS in completely resected stage IB–IIIA NSCLC | Latest news for Doctors, Nurses and Pharmacists | Oncology