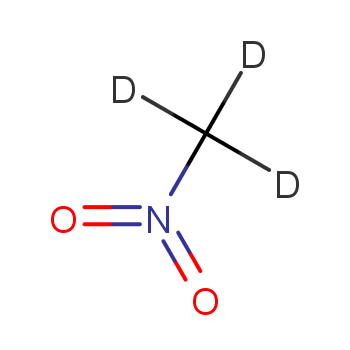
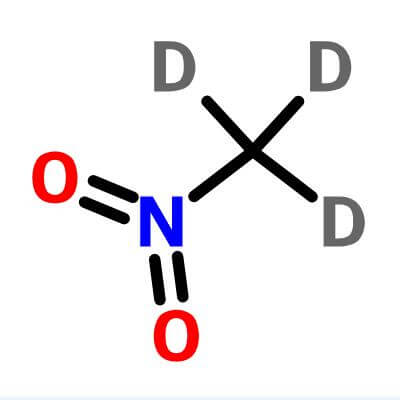
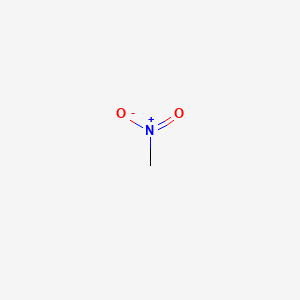
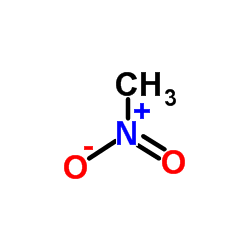
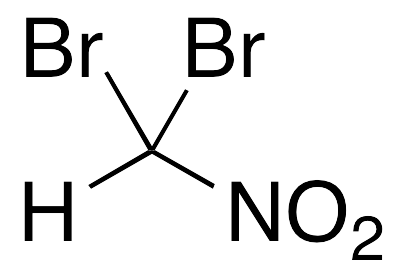R)-chloro(fluoro)nitromethane Structure - CHClFNO2 - Over 100 million chemical compounds | Mol-Instincts

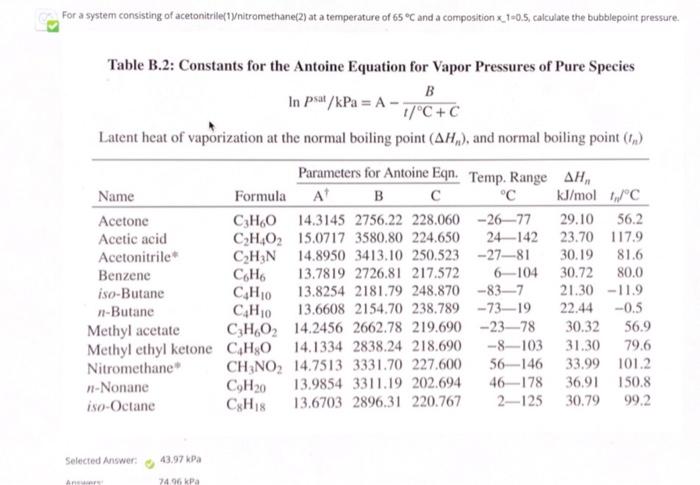
SOLVED: You are given a task by your supervisor to separate acetonitrile from a vapor-phase acetonitrile/nitromethane mixture. The boiling point for nitromethane is higher than acetonitrile. Your colleague suggests to lower the