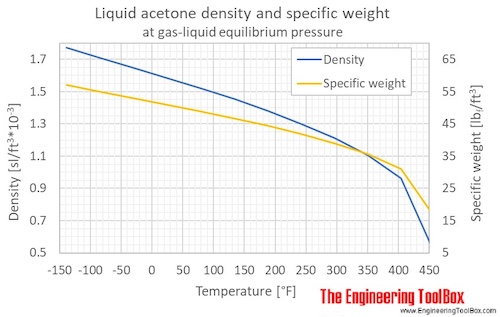
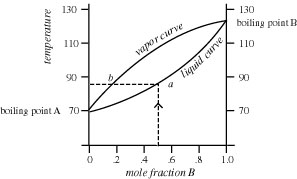
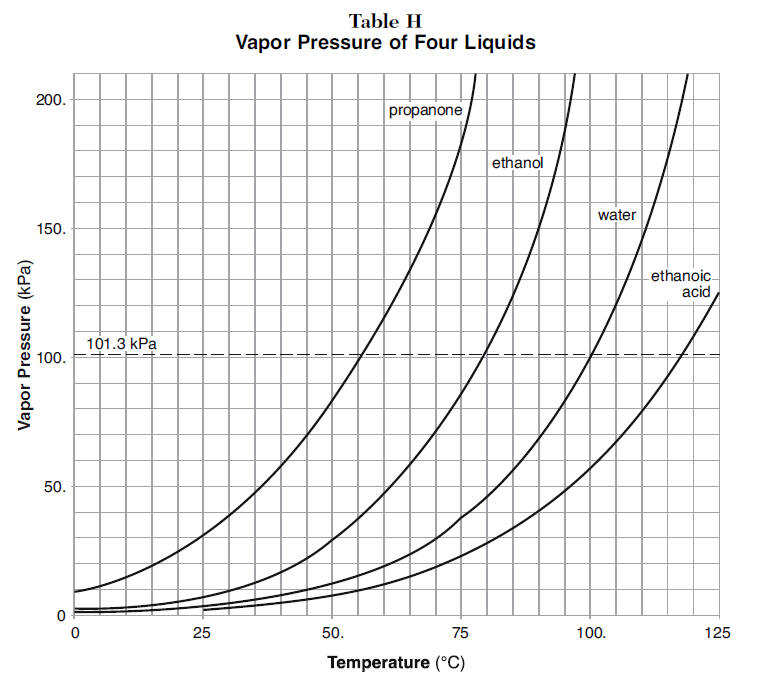
When two volatile liquids of similar boiling points, e.g., acetone `(56^(@)C)` and methanol - YouTube

Enhancement of solvent uptake in porous PVDF nanofibers derived by a water-mediated electrospinning technique - ScienceDirect
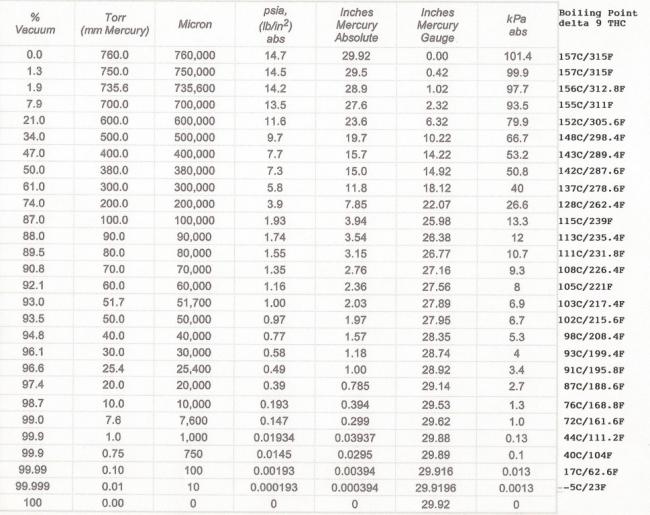
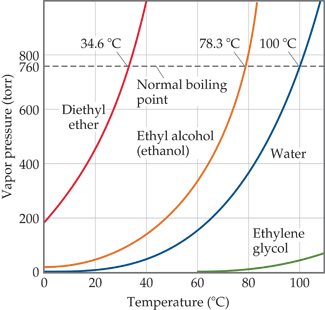
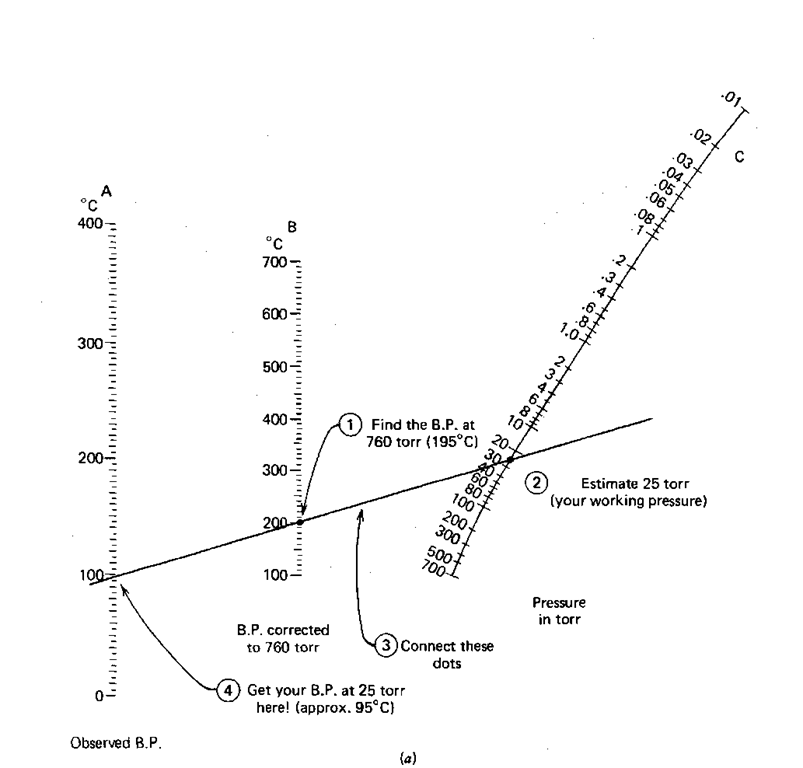
How to calculate the boiling point of acetone in at an atmospheric 630 torr given that the normal boiling point of acetone is 56.5°C - Quora

SOLVED: Which of the following compound pairs could be effectively separated by simple distillation? Briefly explain your choice. sec-butanol and acetophenone aniline and n-hexane isopentane and benzene 0-xylene and acetone Vacuum distillation