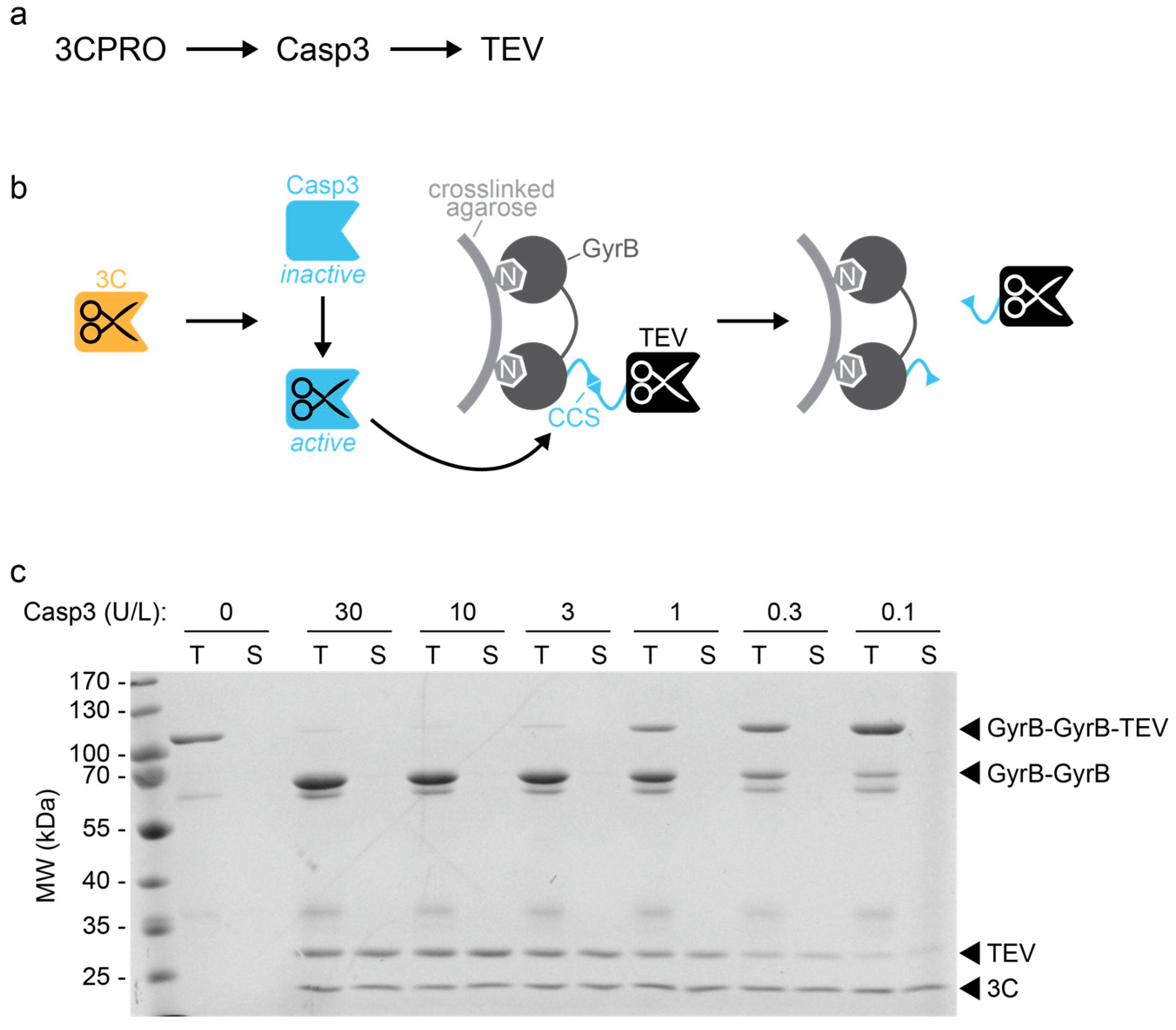
SARS-CoV 3CLpro cleavage sites and the canonical recognition sequence.... | Download Scientific Diagram
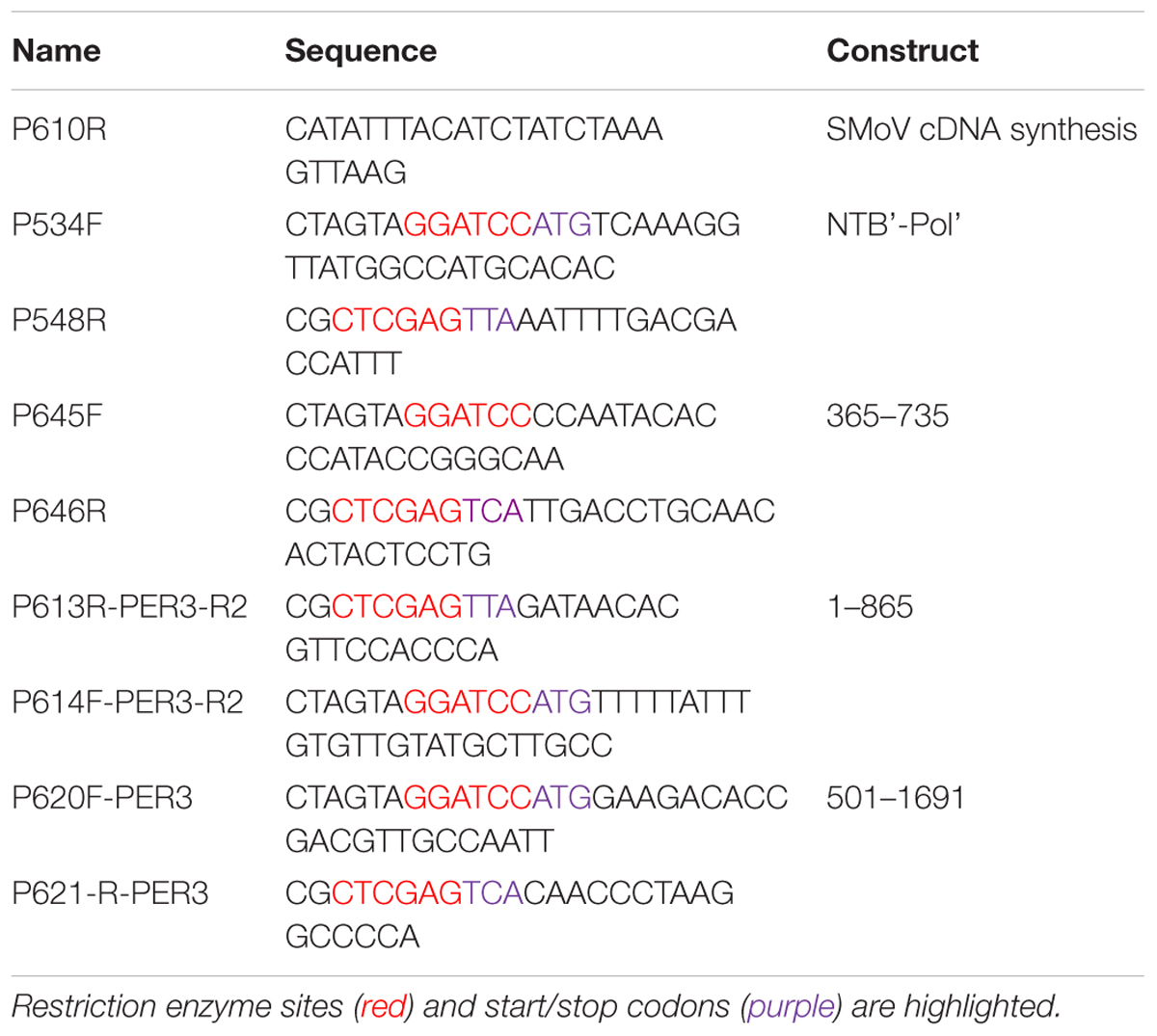
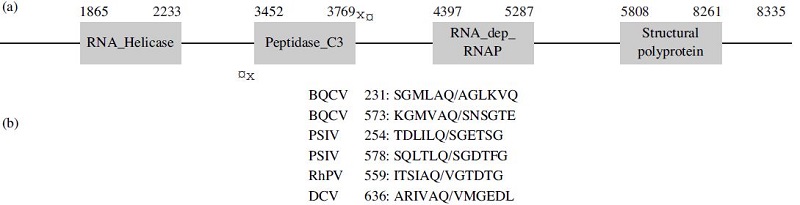
Frontiers | Identification of Cleavage Sites Recognized by the 3C-Like Cysteine Protease within the Two Polyproteins of Strawberry Mottle Virus

Quantitative Analysis of the Substrate Specificity of Human Rhinovirus 3C Protease and Exploration of Its Substrate Recognition Mechanisms | ACS Chemical Biology

Frontiers | Identification of Cleavage Sites Recognized by the 3C-Like Cysteine Protease within the Two Polyproteins of Strawberry Mottle Virus

Figure 2 from and Richard Protease Poly ( A )-Binding Protein by Enterovirus 3 C Efficient Cleavage of Ribosome-Associated | Semantic Scholar
Rhinovirus 3C Protease Facilitates Specific Nucleoporin Cleavage and Mislocalisation of Nuclear Proteins in Infected Host Cells | PLOS ONE

Directed evolution of the 3C protease from coxsackievirus using a novel fluorescence-assisted intracellular method

Frontiers | Running With Scissors: Evolutionary Conflicts Between Viral Proteases and the Host Immune System

Human Rhinovirus 3C protease cleaves RIPK1, concurrent with caspase 8 activation | Scientific Reports

Whole-Genome Sequence Analysis Reveals the Enterovirus D68 Isolates during the United States 2014 Outbreak Mainly Belong to a Novel Clade | Scientific Reports

NT*-HRV3CP: An optimized construct of human rhinovirus 14 3C protease for high-yield expression and fast affinity-tag cleavage - ScienceDirect
Activity of the Human Rhinovirus 3C Protease Studied in Various Buffers, Additives and Detergents Solutions for Recombinant Protein Production | PLOS ONE